ilooda’s Medical Device Wins FDA Approval for Treating Scars After Monkeypox
by Theleaders-Online | July 28, 2022 4:52 am
SEOUL, South Korea, July 28, 2022 /PRNewswire/ — After the first case of monkeypox was reported in the UK in early May, according to data compiled by the US CDC on July 1, 5,783 cases of monkeypox have been reported in 52 countries. Once infected, monkeypox causes headaches along with fever, muscle aches, and fatigue, and it is known to irritate skin through blisters and rashes that later become crusts. In particular, typical sequelae for skin scarring, the degree of inflammation and blisters is significantly greater than from chickenpox, so the scarring is expected to be severe. As a result, dermatologists all over the world who have patients suffering from monkeypox in their country have also started responding with appropriate treatments for monkeypox scarring.
 [1]
[1]
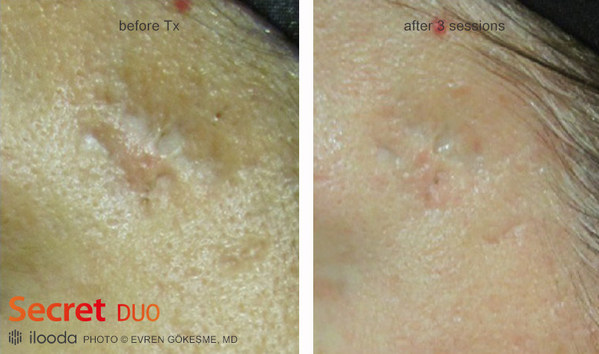
Although various treatments have been introduced to treat scarring from a long time ago, non-isolated microneedle fractional radiofrequency waves are attracting attention as they are highly valued as a treatment device for scarring. Secret Duo of ilooda Co., Ltd., which develops, manufactures and sells specialized medical devices to 70 countries overseas, has now received U.S. FDA approval for its efficacious device for treating scarring. Among the multiple scar treatment modalities, Secret Duo with a fractional radio frequency of 1540 nm laser is an effective treatment of scarring and shows excellent clinical effects.
Secret Duo uses a needle to break excessive fibrous tissue formed in the dermis and promotes collagen synthesis to fill the scar area with new skin. In addition, after fractional radiofrequency treatment, the skin can be improved with a 1540nm laser. The Non-ablative Er:glass 1540 wavelength has a positive effect on scar improvement by stimulating collagen regeneration and causing collagen to contract from water absorption while gently warming healthy skin tissue without thermal damage to the epidermis.
Considering the clinical effect on these scars, a positive effect is expected for the management of scarring from monkeypox infection.
ilooda has recently expanded the cooperation of its academic advisory group with global leading dermatologists and constantly proceeded with the development of state-of-art technology based on clinical expert’s needs and market trends on laser treatment.
![]() View original content to download multimedia:https://www.prnewswire.com/news-releases/iloodas-medical-device-wins-fda-approval-for-treating-scars-after-monkeypox-301594967.html
View original content to download multimedia:https://www.prnewswire.com/news-releases/iloodas-medical-device-wins-fda-approval-for-treating-scars-after-monkeypox-301594967.html
Source: ilooda, Inc.
- [Image]: https://mma.prnasia.com/media2/1864577/image.html
Source URL: https://theleaders-online.com/iloodas-medical-device-wins-fda-approval-for-treating-scars-after-monkeypox/